Common Lancehead |
Reptiles of Ecuador | Serpentes | Viperidae | Bothrops atrox
English common names: Common Lancehead.
Spanish common names: Equis amazónica, equis del oriente (Ecuador); yoperojobobo venenosa (Bolivia); cuatronarices, mapaná, peloegato, talla equis, barba amarilla (Colombia); jergón, jergona, jergón de la selva (Peru); mapanare, rabo amarillo (Venezuela).
Recognition: ♂♂ 135.4 cmMaximum distance from the snout to the tip of the tail. ♀♀ 172.3 cmMaximum distance from the snout to the tip of the tail.. In Amazonian Ecuador, the Common Lancehead (Bothrops atrox) may be identified by having the following combination of features: a triangular-shaped head with a snout that is not upturned, low keels (longitudinal ridges) on the dorsal scales, heat-sensing pits between the eyes and nostrils, and a faint dorsal pattern of 20–33 pale X-shaped markings on a brownish dorsum.1,2 Newly-hatched lanceheads have brightly-colored tail-tips (yellowish in males and whitish in females).3–5 In Ecuador, the most similar viper that may be found living alongside B. atrox is B. brazili, which has a pink iris and a much more contrasting dorsal pattern. The toad-headed pitvipers (genus Bothrocophias) have an upturned snout and comparatively much smaller eyes. The Amazonian Bushmaster (Lachesis muta) differs from B. atrox in having prominent knoblike keels on the dorsal scales, which give the snake a “pineapple texture.”2,5
Picture: Adult from Llanganates National Park,Pastaza, Ecuador. | |
 | |
Picture: Adult from Hato La Aurora, Casanare, Colombia. | |
 | |
Picture: Adult from Hato La Aurora, Casanare, Colombia. | |
 | |
Picture: Juvenile from Maycu Reserve, Zamora Chinchipe, Ecuador. | |
 | |
Natural history: Common, Bothrops atrox is generally the most common viper in the Amazon rainforest.2 It inhabits old-growth to heavily disturbed evergreen lowland and foothill forests, savannas,6 plantations (banana, cassava, African palm,7 and rice fields), pastures, rural gardens, and even ventures into human dwellings.8–10 Individuals are up to 6.4 times more frequently encountered near bodies of water (such as swamps or along lagoons, artificial ponds, rivers, creeks, and streams) than away from them.8,11,12 During “drier” periods in the Amazon (December–February in the Ecuador Amazon), individuals are less frequently encountered and they actively seek wetter spots near bodies of water.12–15
Throughout the day, Common Lanceheads typically remain inactive and coiled on the forest floor (usually along fallen logs, at the base of trees, within buttresses, or in clusters of roots, bamboo clumps, and dense vegetation) or sheltered below leaf-litter, logs and timber. Others remain out in the open,12 basking in direct sunlight or moving at ground level.1–19 Within about an hour of sunset, most individuals emerge from their hideouts and move to their nocturnal ambush sites.4,19 Most individuals remain active until 10:00 pm,12 but others throughout the night.19
Individuals of Bothrops atrox usually dwell on soil or leaf-litter, but also sit-and-wait on the surface of slow-moving bodies of water, swim across rivers, or forage or perch on arboreal vegetation 0.1–4 m above the ground.9,12,20 Overall, there is a tendency for juveniles to be more arboreal than adults.17,21 During a two-day period, one Common Lancehead moved over an area of 43 m2 (about the size of an average studio apartment).19
Common Lanceheads ambush their prey as well as actively forage for it using chemoreception.22,23 They can “bite and hold” their prey or “bite and release,” subsequently following the scent trail of the envenomated prey.14 These vipers are opportunistic predators;24 they feed on almost any animal that is 17–156% of their body size.22,25 As juveniles, they attract prey by means of moving their brightly colored tail as a lure.8,19
The diet of Bothrops atrox includes primarily (38–44%) amphibians (such as Rhinella margaritifera, Boana cinerascens,14 Callimedusa tomopterna,12 Adenomera andreae, Leptodactylus discodactylus,8 L. mystaceus, and L. pentadactylus),18 (19.5–29%) mammals (mostly rodents18,26 but also opossums),10 and (11–19.5%) lizards (such as Arthrosaura reticulata, Loxopholis parietalis, Copeoglossum nigropunctatum, Gonatodes humeralis, Ameiva ameiva, and Kentropyx pelviceps),17,27 but also snakes (including Imantodes cenchoa, Leptodeira annulata, Ninia atrata, Tantilla melanocephala,28 groundsnakes,22 and members of their same species), birds,10 centipedes, and fish.24,29,30 The diet of B. atrox seems to shift from being based primarily on ectothermic (“cold-blooded”) prey as juveniles to based mostly on endothermic (“warm-blooded”) animals as adults.30 Individuals probably obtain water from their prey, from dew-laden surfaces, and bodies of water.
“There are people who believe that the jergón can be vanquished by stealing its venom stash that was temporarily deposited on a leaf by the snake when it drinks water. Many woodsmen swear to me, they have witnessed that when a jergón is thirsty, it will empty its venom glands on a leaf next to the creek and proceed to drink water. While the snake is distracted, the woodsmen would dash in quickly to steal the venom holding leaf. The venom is tossed aside by the woodsman and the snake will whither and die.”
Anthony Giardenelli, American naturalist, 2019.
Common Lanceheads rely on their camouflage as a primary defense mechanism.8 When threatened, some individuals flee, others give a “warning” by vigorously wiggling their tail against the leaf-litter, and some just readily bite if attacked or harassed.4,19 Predators of Bothrops atrox include snakes (such as Clelia clelia25 and Micrurus obscurus),2 mammals (such as peccaries,31 pigs dogs, cats, and tayras), birds (including eagles, hawks, chickens, and trumpeters), caimans (Paleosuchus trigonatus),14 and tarantulas.8,32 Individuals of B. atrox are parasitized by ticks and protozoans.2,14
The Common Lancehead is a venomous species (LD50 1.1–4.9 mg/kg) in which the venom of juveniles is more inflammatory, toxic, hemorrhagic, and kills more quickly than that of adults.33–37 In humans, the venom typically causes intense pain, swelling, bruising, bleeding, blistering, defibrination (depletion of the blood’s coagulation factors), nausea, vomiting, numbness, loss of consciousness, soft-tissue infections, and necrosis (death of tissues and cells).38–41
“A ten-year-old girl in Surinam bitten by a labaria (Bothrops atrox) had her leg bathed in poisonous cassava until the limb grew gangrenous and had to be amputated.”
Dean Ripa, American herpetologist, 2002.25
In poorly managed or untreated cases, the venom of Bothrops atrox can cause amputations, permanent complications and disabilities, and even death (in 1–2% of cases).39–42 The prognosis is usually bad for victims that reach a hospital after six hours after the bite, for patients over 65 or under 15 years old, and for those that use traditional medicine.40,43,44 Patients bitten by neonate snakes are more likely to develop coagulopathy (a condition in which the blood's ability to coagulate is impaired), whereas those bitten by adult snakes are more likely to develop severe local swelling and stronger local tissue damage.45 Critically envenomated victims die from intracranial hemorrhage, acute renal failure, hemorrhagic shock, acute pulmonary oedema (swelling caused by an excess of fluid in body tissues), or respiratory arrest.39,42
“While being captured, two individuals (57.8 and ~100 cm in total length) were able to puncture leather gloves and inject venom on the observer's hand.”
Marcio Martins and Ermelidna Oliveira, Brazilian herpetologists, 1998.4
Bothrops atrox is the most important cause of snakebite mortality in the Amazon.39,46–48 It causes 36.5–99% of snakebites throughout its range,39 probably because snakes of this species are perfectly camouflaged, abundant in agricultural areas, have a high venom yield (up to 342 mg of venom per bite) and toxicity,35,36,49 and have an aggressive self-defense behavior.2
“When he was twelve years old, Don Manuel was bitten by a jergón (Bothrops atrox), a dangerous snake. When he was near death, a young man happened to visit the plantation on which he lived with his family. The young man heard about his case, and said he was going to try an icaro (magic melody) he had learned against snake bites. By singing and blowing smoke over Don Manuel he managed to cure him. Don Manuel learned this icaro during the healing process, and said that he had cured many cases of snake bites during his long life with the help of this magic melody.”
Fortunately, the Colombian and Costa Rican antivenom available in Ecuador can, to a degree, neutralize the venom of Bothrops atrox.42,51 However, the venom’s toxic and enzymatic activities differ drastically between populations, habitat types,52 and across age categories.53–55 For example, the protein similarities between the venom of populations of Common Lanceheads in Brazil and Colombia may only be around 23%.56 Although serum therapy (antivenom) is the only recommended approach against a bite by a B. atrox, the use of dexamethasone as an adjunct to the antivenom therapy could be useful to improve some of the local symptoms caused by the envenomation.57
What to do if you are bitten by a Common Lancehead?
|
Males of Bothrops atrox become sexually mature when they reach ~56.5 cm in total length; females at ~90.6 cm.30 Males fight against each other over access to females; they do so by pushing the opponent to the ground.8,60 The breeding season of some B. atrox populations occurs throughout the year,8,9 but is synchronized with regional rainfall in others.30 Females are capable of delaying fertilization by storing sperm for months.56 After a gestation period of 161–343 (~5–11 months), females “give birth” (the eggs hatch within the mother) to 5–45 young.4,7,8. In captivity, individuals can live up to 18 years.8
Conservation: Least Concern.61 Bothrops atrox is listed in this category because the species is widely distributed, frequently encountered throughout its range, tolerates and even thrives in human-modified environments, and is considered to be facing no major immediate extinction threats.8 However, the Common Lancehead generally cannot survive in areas without vegetation cover. The substitution of traditional crops for mechanized agriculture is causing the species to be less frequently encountered or absent altogether in some areas.14 Other threats include direct killing (lanceheads are usually killed on sight by humans alleging precautionary reasons), traffic mortality, and the decline in the abundance of prey.7 Still, given that B. atrox has formidable capacity to adapt to new environments, it is unlikely that it will become extinct, at least not in the near term future.
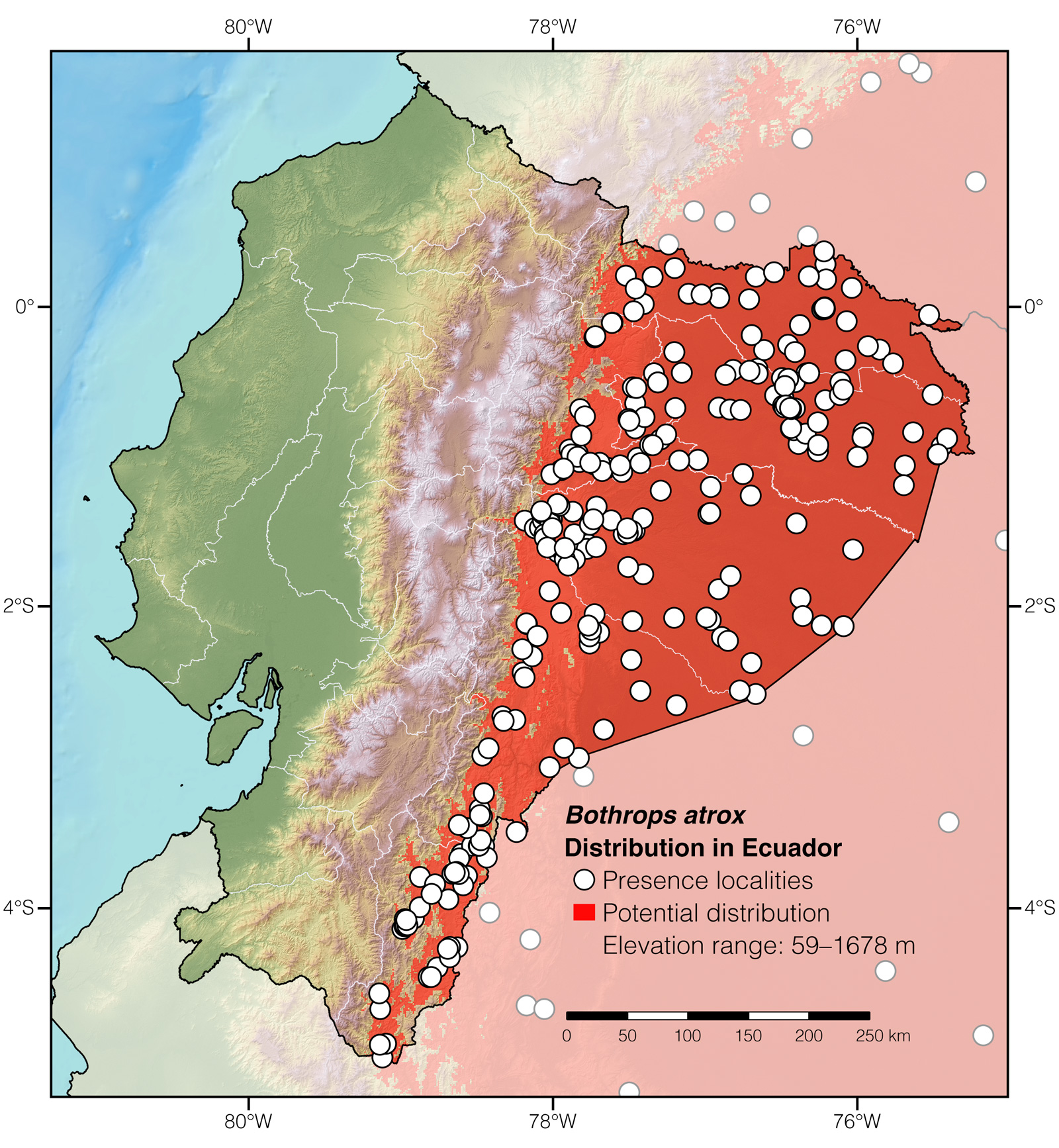
Distribution: Bothrops atrox is native to an estimated ~2,836,368 km2 area throughout the Amazon basin and adjacent foothills of the Andes in Bolivia, Brazil, Colombia, Ecuador, French Guiana, Guyana, Peru, Suriname, and Venezuela.62
Etymology: The generic name Bothrops, which is derived from the Greek word bothros (meaning “pit”),63 refers to the heat-sensing pits between the eyes and nostrils. The specific epithet atrox is a Latin word meaning “savage” or “fierce.” It probably refers to the aggressive self-defense behavior of members of this species.2
Where to observe: Common Lanceheads can be located with ~10–30% certainty in forested or agricultural areas throughout Amazonian Ecuador. Some of the best localities to find Central American Lanceheads in the wild in Ecuador are: Yasuní Scientific Station, Tiputini Biodiversity Station, Jatun Sacha Biological Reserve, Tamandúa Ecological Reserve, Shiripuno Lodge, Copalinga Reseve, and Sani Lodge. The snakes may be located by walking along trails at night.
FREQUENTLY ASKED QUESTIONS: |
Bothrops atrox vs B. asper, which is more dangerous? Although both species of lanceheads are capable of killing a person, the Terciopelo (B. asper) causes more accidents per year,39 has a higher fatality rate (5–7% vs 1–2%),39,64,65 and is generally considered the most dangerous of the two. This is probably because individuals of B. asper attain larger sizes (up to 250.1 cm vs 172.3 cm),2,8 have higher venom yield (up to 1,530 mg vs 342 mg),49,65 and occur in areas with higher human population density. However, the venom of B. atrox is the more toxic of the two (LD50 1.1–4.9 mg/kg vs LD50 1.9–11.2 mg/kg). |
Do vipers follow humans? They don’t, but they follow their prey.22 Vipers such as Bothrops atrox are abundant in human-modified areas because rodents are common in these areas.10 |
Which is the most dangerous snake in the Amazon rainforest? The Common Lancehead (Bothrops atrox) is considered the most dangerous snake in the Amazon, since it causes 36.5–99% of snakebites in the area, and is responsible for the greatest number of fatalities.39 |
Special thanks to Michael Tidwell for symbolically adopting the Common Lancehead and helping bring the Reptiles of Ecuador book project to life.
Click here to adopt a species.
Author: Alejandro ArteagaaAffiliation: Fundación Khamai, Reserva Arlequín, Ecoruta Paseo del Quinde km 56, Santa Rosa de Mindo, Pichincha 171202, Ecuador.
Photographers: Jose VieiraaAffiliation: Tropical Herping (TH), Quito, Ecuador.,bAffiliation: ExSitu, Quito, Ecuador.
How to cite? Arteaga A (2020) Bothrops atrox. In: Arteaga A, Bustamante L, Vieira J (Eds) Reptiles of Ecuador: Life in the middle of the world. Available from: www.reptilesofecuador.com
Literature cited:
- Duellman WE (2005) Cusco amazónico: the lives of amphibians and reptiles in an Amazonian rainforest. Cornell University Press, Ithaca, 433 pp.
- Campbell JA, Lamar WW (2004) The venomous reptiles of the western hemisphere. Cornell University Press, Ithaca, 774 pp.
- Natera-Mumaw M, Esqueda-González LF, Castelaín-Fernández M (2015) Atlas serpientes de Venezuela. Dimacofi Negocios Avanzados S.A., Santiago de Chile, 456 pp.
- Martins M, Oliveira ME (1998) Natural history of snakes in forests of the Manaus region, Central Amazonia, Brazil. Herpetological Natural History 6: 78–150.
- Harvey MB, Aparicio J, Gonzales L (2009) Revision of the venomous snakes of Bolivia. II. The pitvipers (Serpentes: Viperidae). Annals of the Carnegie Museum 74: 1–37.
- Farias RES, Rocha AM, Viana PF (2019) Bothrops atrox (Common Lancehead). Habitat use. Herpetological Review 50: 585–586.
- Quiñones-Betancourt E, Díaz-Ricaurte JC, Angarita-Sierra T, Guevara-Molina EC, Díaz-Morales RD (2018) Bothrops atrox (Linnaeus, 1758). Catálogo de Anfibios y Reptiles de Colombia 4: 7–23.
- Valencia JH, Garzón-Tello K, Barragán-Paladines ME (2016) Serpientes venenosas del Ecuador: sistemática, taxonomía, historial natural, conservación, envenenamiento y aspectos antropológicos. Fundación Herpetológica Gustavo Orcés, Quito, 653 pp.
- Duellman WE (1978) The biology of an equatorial herpetofauna in Amazonian Ecuador. Publications of the Museum of Natural History, University of Kansas 65: 1–352.
- Cunha OR, Nascimento FP (1975) Ofídios da Amazônia. VII—As serpentes peçonhentas do gênero Bothrops (jararacas) e Lachesis (surucucu) da região leste do Pará (Ophidia, Viperidae). Boletim do Museu Paraense Emílio Goeldi 83: 1–42.
- de Fraga R, Magnusson WE, Abrahão CR, Sanaiotti T, Lima AP (2013) Habitat selection by Bothrops atrox (Serpentes: Viperidae) in Central Amazonia, Brazil. Copeia 2013: 684–690.
- Oliveira ME, Martins M (2001) When and where to find a pitviper: activity patterns and habitat use of the lancehead, Bothrops atrox, in central Amazonia, Brazil. Herpetological Natural History 8: 101–110.
- Cunha OR, Nascimento FP (1993) Ofídios da Amazônia. As cobras da região leste do Pará. Papéis Avulsos Museu Paraense Emílio Goeldi 40: 9–87.
- Field notes, Reptiles of Ecuador book project.
- Henderson RW, Dixon JR, Soini P (1978) On the incidence of tropical snakes. Milwaukee Public Museum Contributions in Biology and Geology 17: 1–15.
- Emsley M (1977) Snakes, and Trinidad and Tobago. Bulletin of the Maryland Herpetological Society 13: 201–304.
- Dixon JR, Soini P (1986) The reptiles of the upper Amazon Basin, Iquitos region, Peru. Milwaukee Public Museum, Milwaukee, 154 pp.
- Beebe W (1946) Field notes on the snakes of Kartabo, British Guiana, and Caripito, Venezuela. Zoologica 31: 11–52.
- Henderson RW, Nickerson MA, Ketcham S (1976) Short term movements of the snakes Chironius carinatus, Helicops angulatus and Bothrops atrox in Amazonian Peru. Herpetologica 32: 304–310.
- Melo-Sampaio PR, Lima Maciel JM (2018) Bothrops atrox (Common Lancehead, Jararaca). Habitat use. Herpetological Review 49: 124.
- Turci LCB, Albuquerque S, Bernarde PS, Miranda DB (2009) Uso do hábitat, atividade e comportamento de Bothriopsis bilineatus e de Bothrops atrox (Serpentes: Viperidae) na floresta do Rio Moa, Acre, Brasil. Biota Neotropica 9: 197–206.
- Egler SG, Oliveira ME, Martins M (1996) Bothrops atrox (Common lancehead). Foraging behavior and ophiophagy. Herpetological Review 27: 22–23.
- Martins M, Marques OAV, Sazima I (2002) Ecological and phylogenetic correlates of feeding habits in Neotropical pitvipers of the genus Bothrops. In: Schuett GW, Höggren M, Douglas ME, Greene HW (Eds) Biology of the vipers. Eagle Mountain Publishing, Eagle Mountain, 307–328.
- Oliveira ME, Martins M (2003) Bothrops atrox (Common Lancehead). Prey. Herpetological Review 34: 61–62.
- Ripa D (2002) The bushmasters: morphology in evolution and behavior. Cape Fear Serpentarium, Wilmington, 349 pp.
- Moldowan PD (2016) Bothrops atrox (Common Lancehead). Diet and fang replacement. Herpetological Review 47: 473–474.
- Avila-Pires TCS (1995) Lizards of Brazilian Amazonia (Reptilia: Squamata). Zoologische Verhandelingen 299: 1–706.
- Murphy JC (1997) Amphibians and reptiles of Trinidad and Tobago. Krieger, Malabar, 245 pp.
- Martins M, Gordo M (1993) Bothrops atrox (Common Lancehead). Diet. Herpetological Review 24: 151–152.
- Bisneto PF, Kaefer IL (2019) Reproductive and feeding biology of the common lancehead Bothrops atrox (Serpentes, Viperidae) from central and southwestern Brazilian Amazonia. Acta Amazonica 49: 105–113.
- Sasa M, Wasko DK, Lamar WW (2009) Natural history of the Terciopelo Bothrops asper (Serpentes: Viperidae) in Costa Rica. Toxicon 54: 904–922.
- Almeida MQ, Sobral R, Da Silva-Neto AM, Mendes DMM, Hidalgo RM (2019) Bothrops atrox (Common Lancehead). Predation. Herpetological Review 50: 586.
- Saldarriaga MM, Otero R, Núñez V, Toro MF, Díaz A, Gutiérrez JM (2003) Ontogenetic variability of Bothrops atrox and Bothrops asper snake venoms from Colombia. Toxicon 42: 405–411.
- Sanchez EF, Freitas TV, Ferreira-Alves DL, Velarde DT, Diniz MR, Cordeiro MN, Agostini-Cotta G, Diniz CR (1992) Biological activities of venoms from South American snakes. Toxicon 30: 95–103.
- Kuch U, Mebs D, Gutiérrez JM, Freire A (1996) Biochemical and biological characterization of the Ecuadorian pitvipers (genera Bothriechis, Bothriopsis, Bothrops, and Lachesis). Toxicon 34: 714–717.
- Terán MC, Lomonte B (2016) Actividad letal de seis venenos de serpientes de importancia médica en el Ecuador. Revista Ecuatoriana de Medicina y Ciencias Biológicas 37: 25–30.
- Laing GD, Yarleque A, Marcelo A, Rodríguez, E., Warrell DA, Theakston RDG (2004) Preclinical testing of three south American antivenoms against the venom of five medically-important Peruvian snake venoms. Toxicon 44: 103–106.
- Rojas E, Quesada L, Arce V, Lomonte B, Rojas G, Gutiérrez JM (2005) Neutralization of four Peruvian Bothrops sp. snake venoms by polivalent antivenoms produced in Perú and Costa Rica: preclinical assessment. Acta Tropica 93: 85–95.
- Warrell DA (2004) Snakebites in Central and South America: epidemiology, clinical features, and clinical management. In: Campbell JA, Lamar WW (Eds) The Venomous reptiles of the Western Hemisphere. Cornell University Press, Ithaca, 709–761.
- Sachett JAG, Val FF, Alcantara JA, Cubas-Vega N, Montenegro CS, da Silva IM, de Souza TG, Santana MF, Ferreira LCL, Monteiro WM (2020) Bothrops atrox snakebite: how a bad decision may lead to a chronic disability: a case report. Wilderness & Environmental Medicine.
- Sant’Ana Malaque CM, Gutiérrez JM (2015) Snakebite envenomation in Central and South America. In: Brent J, Burkhart K, Dargan P, Hatten B, Mégarbane B, Palmer R, White J (Eds) Critical care toxicology. Springer, Basel, 1–22.
- Smalligan R, Cole J, Brito N, Laing GD, Mertz BL, Manock S, Maudlin J, Quist B, Holland G, Nelson S, Lalloo DG, Rivadeneira G, Barragán ME, Dolley D, Eddleston M, Warrell DA, Theakston RDG (2004) Crotaline snake bite in the Ecuadorian Amazon: randomised double blind comparative trial of three South American polyspecific antivenoms. BMJ 329: 1129–1135.
- Borges CC, Sadahiro M, dos Santos MC (1999) Aspectos epidemiológicos e clínicos dos acidentes ofídicos ocorridos nos municípios do Estado do Amazonas. Revista da Sociedade Brasileira de Medicina Tropical 32: 637–646.
- Feitosa EL, Sampaio VS, Salinas JL, Queiroz AM, da Silva IM, Gomes AA, Sachett J, Siqueira AM, Ferreira LC, Dos Santos MC, Lacerda M, Monteiro W (2015) Older age and time to medical assistance are associated with severity and mortality of snakebites in the Brazilian Amazon: a case-control study. PLoS ONE 10: e0132237.
- Bernal JCC, Bisneto PF, Pereira JPT, Ibiapina H, Sarraff LKS, Monteiro-Junior C, da Silva Pereira H, Santos B, de Moura VM, de Oliveira SS, Lacerda M, Sampaio V, Kaefer IL, Gutierrez JM, Bernarde PS, Fan HW, Sachett J, da Silva AMM, Monteiro WM (2020) “Bad things come in small packages”: predicting venom-induced coagulopathy in Bothrops atrox bites using snake ontogenetic parameters. Clin Toxicol (Phila) 58: 388-396.
- Zavaleta A, Salas M (1996) Ofidismo: envenenamiento por mordedura de serpientes. In: Martínez-Villaverde JR, León-Barúa R, Vidal-Neira L, Losno-García R (Eds) Emergencias en medicina interna. Boehringer, Lima, 241–260.
- dos Santos MC (2003) Serpentes peconhentas e ofidismo no Amazonas. In: Cardoso JLC, França FOS, Wen FH, Málaque CMS, Haddad Jr V (Eds) Animais peconhentos no Brasil. FAPESP, Sao Paulo, 115–125.
- Pardal PPO, Monteiro MRCC, Arnaund RN, Lopes FOB, Asano ME (1995) Aspectos epidemiológicos de 465 acidentes ofídicos atendidos no HUJBB - Belém - Pará, no período de 1993–1994. Revista da Sociedade Brasileira de Medicina Tropical 28: 1–170.
- Rosenfeld G (1971) Symptomatology, pathology, and treatment of snake bites in South America. In: Bücherl W, Buckley EE (Eds) Venomous animals and their venoms. Academic Press, New York, 345–384.
- Luna-Medina LE (1992) Icaros: magic melodies among the mestizo shamans of the Peruvian Amazon. In: Matteson EJ, Baer G (Eds) Portals of power: shamanism in South America. University of New Mexico Press, Albuquerque, 231–253.
- Núñez V, Cid P, Sanz L, De La Torre P, Angulo Y, Lomonte B, Gutiérrez JM, Calvete JJ (2009) Snake venomics and antivenomics of Bothrops atrox venoms from Colombia and the Amazon regions of Brazil, Perú and Ecuador suggest the occurrence of geographic variation of venom phenotype by a trend towards paedomorphism. Journal of Proteomics 73: 57–78.
- Sousa LF, Portes-Junior JA, Nicolau CA, Bernardoni JL, Nishiyama MY, Jr., Amazonas DR, Freitas-de-Sousa LA, Mourao RH, Chalkidis HM, Valente RH, Moura-da-Silva AM (2017) Functional proteomic analyses of Bothrops atrox venom reveals phenotypes associated with habitat variation in the Amazon. J Proteomics 159: 32-46.
- Guércio RAP, Shevchenko A, Shevchenko A, López-Lozano JL, Paba J, Sousa MV, Ricart CAO (2006) Ontogenetic variations in the venom proteome of the Amazonian snake Bothrops atrox. Proteome Science 4: 11.
- López-Lozano JL, Sousa MV, Ricart CAO, Cháves-Olortegui C, Sanchez EF, Muniz EG, Bührnheim PF, Morhy L (2002) Ontogenetic variation of metalloproteinases and plasma coagulant activity in venoms of wild Bothrops atrox specimens from Amazonian rain forest. Toxicon 40: 997–1006.
- Cavinato RA, Remold H, Kipnis TL (1998) Purification and variability in thrombin-like activity of Bothrops atrox venom from different geographic regions. Toxicon 36: 257–267.
- Field notes of Darwin Núñez.
- Santos Barreto GN, de Oliveira SS, Dos Anjos IV, Chalkidis HM, Mourao RH, Moura-da-Silva AM, Sano-Martins IS, Goncalves LR (2017) Experimental Bothrops atrox envenomation: efficacy of antivenom therapy and the combination of Bothrops antivenom with dexamethasone. PLoS Neglected Tropical Diseases 11: e0005458.
- Hardy DL (1994) Bothrops asper (Viperidae) snakebite and field researchers in Middle America. Biotropica 26: 198–207.
- Avau B, Borra V, Vandekerckhove P, De Buck E (2016) The treatment of snake bites in a first aid setting: a systematic review. PLoS Neglected Tropical Diseases 10: e0005079.
- Shine R (1994) Sexual size dimorphism in snakes revisited. Copeia 1994: 326–346.
- Carrillo E, Aldás A, Altamirano M, Ayala F, Cisneros-Heredia DF, Endara A, Márquez C, Morales M, Nogales F, Salvador P, Torres ML, Valencia J, Villamarín F, Yánez-Muñoz M, Zárate P (2005) Lista roja de los reptiles del Ecuador. Fundación Novum Millenium, Quito, 46 pp.
- Nogueira CC, Argôlo AJS, Arzamendia V, Azevedo JA, Barbo FE, Bérnils RS, Bolochio BE, Borges-Martins M, Brasil-Godinho M, Braz H, Buononato MA, Cisneros-Heredia DF, Colli GR, Costa HC, Franco FL, Giraudo A, Gonzalez RC, Guedes T, Hoogmoed MS, Marques OAV, Montingelli GG, Passos P, Prudente ALC, Rivas GA, Sanchez PM, Serrano FC, Silva NJ, Strüssmann C, Vieira-Alencar JPS, Zaher H, Sawaya RJ, Martins M (2019) Atlas of Brazilian snakes: verified point-locality maps to mitigate the Wallacean shortfall in a megadiverse snake fauna. South American Journal of Herpetology 14: 1–274.
- Brown RW (1956) Composition of scientific words. Smithsonian Books, Washington D.C., 882 pp.
- Otero R, Osorio RG, Valderrama R, Giraldo CA (1992) Efectos farmacológicos y enzimáticos de los venenos de serpientes de Antioquia y Chocó (Colombia). Toxicon 30: 611–620.
- Bolaños RA (1982) Las serpientes venenosas de Centro América y el problema del ofidismo. Aspectos zoológicos, epidemiológicos y biomédicos. Revista Costarricense de Ciencias Médicas 3: 165–184.